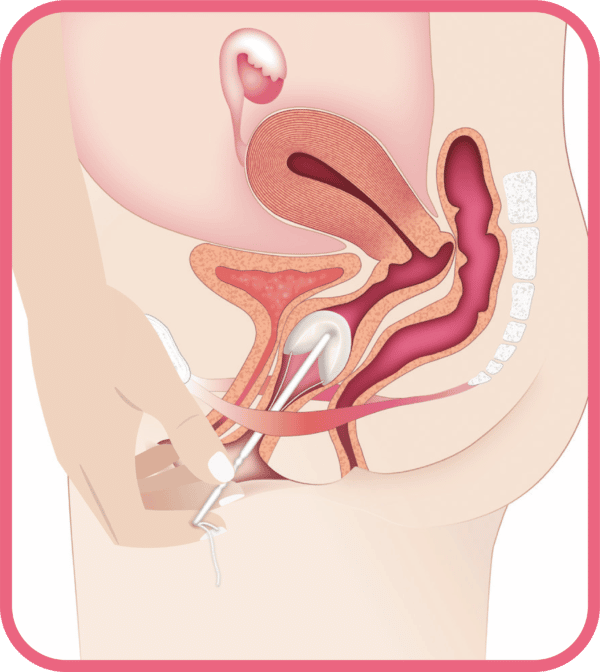
Contraindications
You should not use Contrelle if you:
● are under the age of 18 years
● are pregnant, or have given birth in the last 3 months
● are suffering from any infection or irritation in the vagina
● have suffered from TSS (Toxic Shock Syndrome)
● have had any pelvic surgery in the last 6 months
● are undergoing any therapy for pelvic malignancy
● have moderate to severe vaginal prolapse
● have urinary or vaginal infections
● have significant or bothersome vaginal dryness
● have problems with manual dexterity which could cause insertion or removal issues
● are male
Hazards and Cautions
Do not use the product more than once.
Single use device not to be used for more than 16 hours in any 24 hour period or for more than 6 hours during menstruation. See IFU for full contraindications and cautions.
Ingredients
Biocompatible foam
Clinical Studies
1. Effect of a Vaginal Device on Quality of Life With Urinary Stress Incontinence Pia Sander, MD, Hans Thyssen, MD, Gunnar Lose, MD, DMSc AND Jens Thorup Andersen MD, DMSc Obstetrics & Gynaecology, Vol 93, 3 March 1999
2. Treatment of female stress urinary incontinence with a new anatomically shaped vaginal device (Conveen Continence Guard) I Hahn & I. Milsom British Journal of Urology, Volume 77 Number 5 Pages 711-715
3. Effect of vaginal devices on bladder neck mobility in stress incontinent women Lone Mouritsen, Department of Obstretrics and Gynecology, Gentofte Hospital, University of Copenhagen, Denmark Acta Obstetricia et Gynecologica Scandinavica 2001 80: 428-431
4. New disposable vaginal device (continence guard) in the treatment of female stress incontinence: Design, efficacy and short term safety Hans Thyssen and Gunnar Lose, Glostrup County Hospital, University of Copenhagen, Denmark Acta Obstetricia et Gynecologica Scandinavica 1996: 75 -173
5. Long Term Efficacy and Safety of a Disposable Vaginal Device (Continence Guard) in the Treatment of Female Stress Incontinence H. H Thyssen and G. Lose, Glostrup County Hospital, University of Copenhagen, Denmark International Urogynaecology Journal 1997 8:130-133
6. A new intravaginal device for stress incontinence in women H Thyssen, J. Bidemead, g Lose, K Moller Bek, P Dwyer, L Cardozo Glostrup County Hospital, University of Copenhagen, Denmark, Kings College Hospital London, Skejby Hospital Denmark, The Royal Women’s Hospital Melbourne Australia BJU Int. 2001 Dec 88:889-92
7. A Vaginal Device (Continence Guard) in the Management of Urge Incontinence in women H.Thyssen, P. Sander, G.Lose Int. Urogynaecology Journal 1999 10:219-222
|